Scientists track evolution of microbes on the skin’s surface
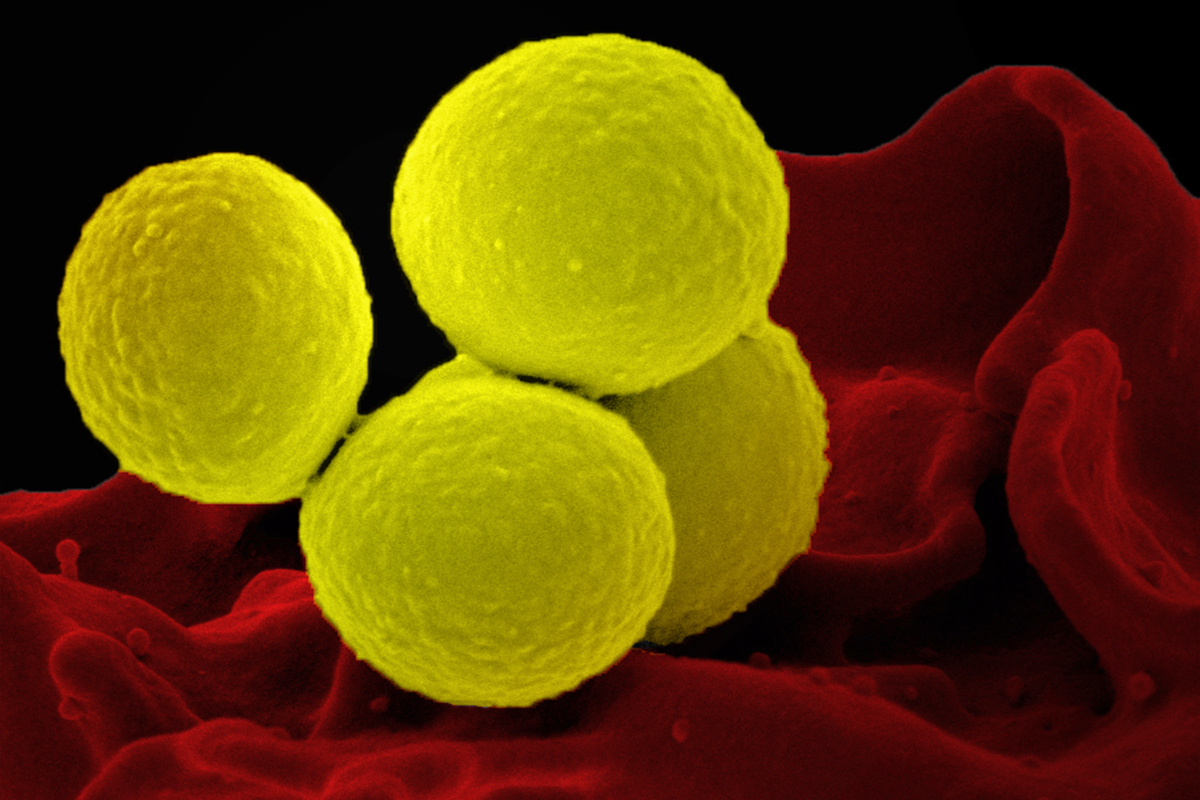
Human skin is home to millions of microbes. One of these microbes, Staphylococcus aureus, is an opportunistic pathogen that can invade patches of skin affected by eczema, also known as atopic dermatitis.
In a new study, researchers at MIT and other institutions have discovered that this microbe can rapidly evolve within a single person’s microbiome. They found that in people with eczema, S. aureus tends to evolve to a variant with a mutation in a specific gene that helps it grow faster on the skin.
This study marks the first time that scientists have directly observed this kind of rapid evolution in a microbe associated with a complex skin disorder. The findings could also help researchers develop potential treatments that would soothe the symptoms of eczema by targeting variants of S. aureus that have this type of mutation and that tend to make eczema symptoms worse.
“This is the first study to show that Staph aureus genotypes are changing on people with atopic dermatitis,” says Tami Lieberman, an assistant professor of civil and environmental engineering and a member of MIT’s Institute for Medical Engineering and Science. “To my knowledge, this is the most direct evidence of adaptive evolution in the skin microbiome.”
Lieberman and Maria Teresa García-Romero, a dermatologist and assistant professor at the National Institute of Pediatrics in Mexico, are the senior authors of the study, which appears today in Cell Host and Microbe. Felix Key, a former MIT postdoc who is now a group leader at the Max Planck Institute for Infection Biology, is the lead author of the paper.
Bacterial adaptation
It is estimated that between 30 and 60 percent of people carry S. aureus in their nostrils, where it is usually harmless. In people with eczema, which affects about 10 million children and 16 million adults in the United States, S. aureus often spreads to eczema patches and infects the skin.
“When there’s a break in the skin, Staph aureus can find a niche where it can grow and replicate,” Lieberman says. “It’s thought that the bacteria contribute to the pathology because they secrete toxins and recruit immune cells, and this immune reaction further damages the skin barrier.”
In this study, the researchers wanted to explore how S. aureus is able to adapt to living on the skin of eczema patients.
“These microbes normally live in the nose, and we wanted to know whether when it finds itself on atopic dermatitis skin, does it need to change to live there? And can we learn something about how these bacteria are interacting with atopic dermatitis skin from watching its evolution?” Lieberman says.
To answer those questions, the researchers recruited patients aged 5 to 15 who were being treated for moderate to severe eczema. They took samples of the microbes on their skin once a month for three months, and then again at nine months. Samples were taken from the backs of the knees and inside of the elbows (the most common sites affected by eczema), the forearms, which are usually not affected, and the nostrils.
S. aureus cells from each sample site were cultured separately to create up to 10 colonies from each sample, and once large colonies formed, the researchers sequenced the cells’ genomes. This yielded nearly 1,500 unique colonies, which enabled the researchers to observe the bacterial cells’ evolution in much greater detail than has previously been possible.
Using this technique, the researchers found that most patients maintained a single lineage of S. aureus — that is, it was very uncommon for a new strain to come in from the environment or another person and replace the existing S. aureus strain. However, within each lineage, a great deal of mutation and evolution occurred during the nine months of the study.
“Despite the stability at the lineage level, we see a lot of dynamics at the whole genome level, where new mutations are constantly arising in these bacteria and then spreading throughout the entire body,” Lieberman says.
Many of these mutations arose in a gene called capD, which encodes an enzyme necessary for synthesizing the capsular polysaccharide — a coating that protects S. aureus from recognition by immune cells. In two out of six deeply sampled patients, cells with capD mutations took over the entire S. aureus skin microbiome population, the researchers found. Other patients were colonized with strains initially lacking a functional copy of the capD, for a total of 22 percent of patients lacking capD at the end of the study. In one patient, four different mutations of capD arose independently in different S. aureus samples, before one of those variants became dominant and spread over the entire microbiome.
Targeted treatment
In tests of bacterial cells growing in a lab dish, the researchers showed that mutations to capD allowed S. aureus to grow faster than S. aureus strains with a normal capD gene. Synthesizing the capsular polysaccharide requires a lot of energy, so when cells don’t have to make it, they have more fuel to power their own growth. The researchers also hypothesize that loss of the capsule may allow the microbes to stick to the skin better because proteins that allow them to adhere to the skin are more exposed.
The researchers also analyzed nearly 300 publicly available genomes of bacteria isolated from people with and without eczema, and found that people with eczema were much more likely to have S. aureus variants that could not produce the capsular polysaccharide than people without eczema.
Eczema is usually treated with moisturizers or topical steroids, and doctors may prescribe antibiotics if it appears that the skin is infected. The researchers hope that their findings could lead to the development of treatments that lessen eczema symptoms by targeting S. aureus variants with mutations in the capsular polysaccharide.
“Our findings in this study provide clues as to how Staph aureus is evolving inside hosts and reveal some of the features that might help the bacteria to stay on the skin and generate disease versus being able to be swiped off,” García-Romero says. “In the future, S. aureus variants with mutations in the capsular polysaccharide could be a relevant target for potential treatments.”
Lieberman’s lab is now working on developing probiotics that could be used to target the capsule-negative S. aureus strains. Her lab is also studying whether S. aureus strains with capD mutations are more likely to spread to other members of an eczema patient’s household.
The research was funded by MISTI Global Seed Funds, the U.S. National Institutes of Health, the Burroughs Wellcome Fund, the Mexican Government Ministry of Taxes Program for Health Research and Technological Development, and a German Research Foundation fellowship.
Share on Bluesky


